Oxford university started the first human vaccine trial in the United Kingdom to fight against novel coronavirus. They injected the vaccine into two volunteers, the first of over eight hundred individuals recruited for the study. Half of these volunteers will receive the vaccine of COVID-19, while half will receive a control vaccine that defends against meningitis but not pandemic. Previously, last week Oxford University announced to start Coronavirus vaccine human trials.
The first human trial in Europe of a #coronavirus vaccine has begun in Oxford. Two volunteers were injected, the first of more than 800 people recruited for the study. https://t.co/QvuNFus42Q https://t.co/0SpVXgFJip
— Rachael Akidi (@rakidi) April 24, 2020
The design of the vaccine trial means that these patients will not know the vaccine name they are getting; however, doctors will. One of the two individuals who received the vaccine injection, Elisa Granato told a news reporter that as a scientist he wanted to stab to back the scientific process whenever he can.
First Coronavirus Vaccine Developed in under 3 months
A team at Oxford University developed the vaccine in under three months. Professor of vaccinology at the Jenner Institute, Sarah Gilbert, commanded the pre-clinical research. She showed her high level of confidence in the success of the vaccine.
The #vaccine was developed in under three months by a team at #OxfordUniversity. Sarah Gilbert, professor of vaccinology at the Jenner Institute, led the pre-clinical research.https://t.co/oIcgQIV4jJ
— DT Next (@dt_next) April 24, 2020
Furthermore, she expressed that they have to test the vaccine and get information from humans. The team of scientists needs to reveal that vaccine actually works and stops more individual getting infected with COVID-19 before using it in the broader population.
Previously, professor Gilbert showed her eighty percent confidence in the success of the vaccine, but this time she preferred not to put any figure about it and expressed generally her optimism about its chances.
How Does the Vaccine will work?
The scientist made the vaccine from a debilitated version of a usual cold virus (known as an adenovirus) from chimpanzees. They modified it so it can’t grow in humans.
Scientists took the genes for the spike protein on the coronavirus surface and put them into a harmless virus to make a vaccine.
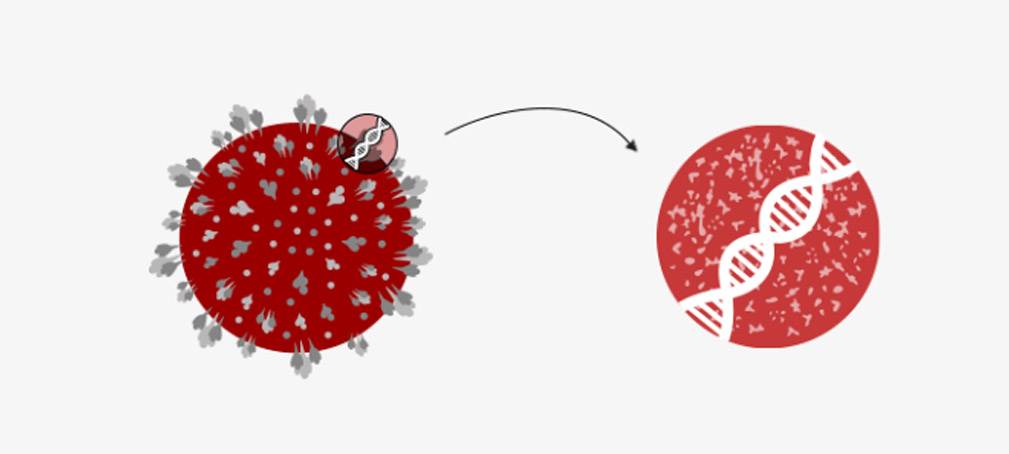
Now, the vaccine injected into the patient
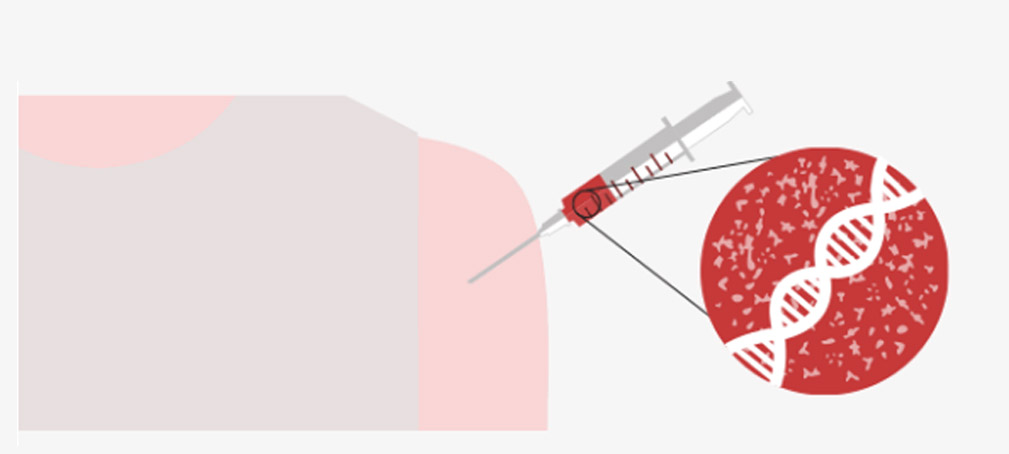
The vaccine enters cells, which then began to generate the COVID-19 spike protein. It prompts the human immune system to produce antibodies and activate killer T-cells to destroy infected cells.
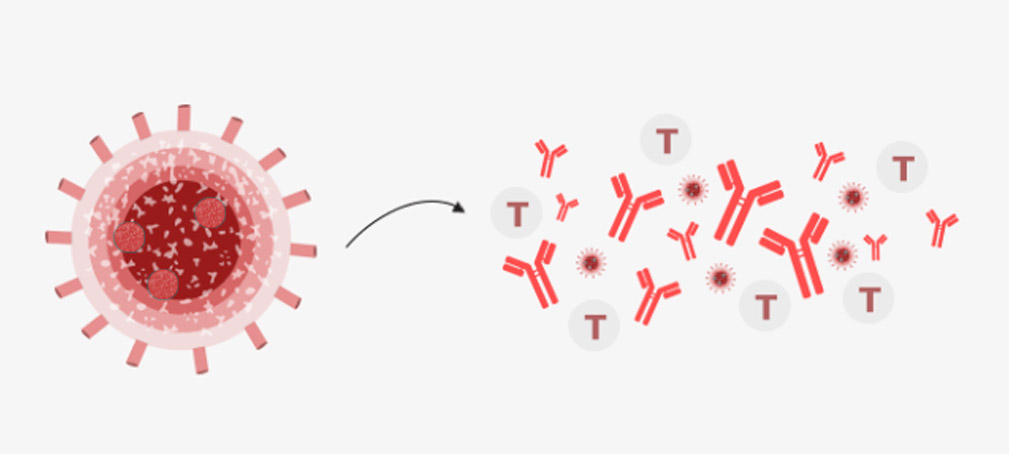
If the patient encounters COVID-19 again, the T cells and antibodies triggered to fight the virus
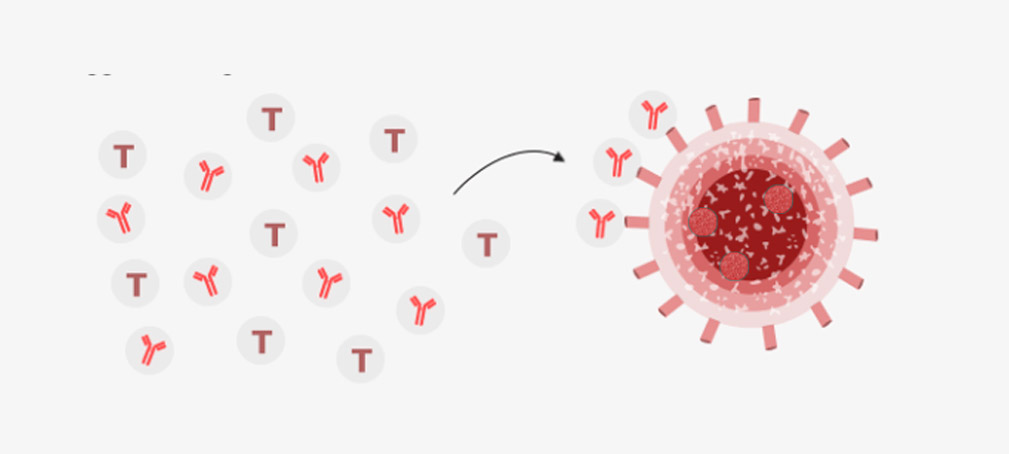
The Oxford scientist’s team already made a vaccine against Mers, another coronavirus type, using the same approach – and that received promising results in clinical trials.
How will scientists know if it works?
The only way, the team will know if the coronavirus vaccine works are by relating the number of individuals infected with the virus in the months before from the two arms of the trial. That may be a matter of problem if a number of cases fall quickly in the United Kingdom, because in this case, they may not collect enough information.
The director of the Oxford Vaccine Group, Professor Andrew Pollard, is leading the trial. He adds that they are chasing the end of this current coronavirus wave. If they do not clasp that, they will not be able to tell whether the COVID-19 vaccine works in the upcoming few months. However, they have an expectation that there will be further cases in the coming few months because this virus has not gone away.
The researchers of the vaccine are prioritizing the enrollment of local healthcare workers to take their services for the trial process because they are more likely to expose to the virus than others. A larger trial of around five thousand volunteers will start in the future for a few months and will have no age limit. As it is a common practice that older people tend to have somewhat weaker immune responses to vaccines. Researchers are working on this scenario, whether they may need two doses of the vaccine.
The team of Oxford is also working in collaboration with the researchers in Kenya about a probable vaccine there, where the transmission rates are mounting from a lower base. If the infected numbers might a problem, why not intentionally infect volunteers with the COVID-19. That might be an instant and firm way to determine if the vaccine’s results became effective. Whereas, it might be morally questionable because of no approved treatments for coronavirus.
Is this method safe?
The volunteers of the trial would wisely observe in the coming months. They said that some volunteers might take a sore arm, fevers, or headaches during the first few days after the vaccination. According to the Oxford team, the statistics suggest that the risk of vaccine creating a higher disease is negligible, and the figures from the studies in animals are positive and satisfactory. Scientists are hopeful that by September 2020, they will ready around one million doses and will dramatically expand manufacturing after that.
Read Also: Coronavirus causes sudden strokes in youngsters – health experts